Morbidity and Mortality rounds (M&M rounds) provide a forum for medical education, quality improvement, and risk management. They enhance the ability of medical staff to participate in clinical care quality improvement and bring together the multidisciplinary team to learn as a team and to escalate improvement ideals beyond their sphere of influence, all of which contribute to improved patient care quality and safety in Island Health.

M&M rounds are to align with the organizational values of Courage, Aspiration, Respect, and Empathy and are guided by the following principles:
- Shared accountability for quality across the organization and between professions.
- Shared information relating to quality to promote learning and spread of good practice.
- Timeliness and responsiveness, recognizing that matters presenting an urgent threat to safety are expedited, signaling quality and safety as a top priority in Island Health.
- A culture of continuous improvement and psychologically safe learning environments.
Attendance at M&M rounds can be claimed as a Continuing Medical Education (CME) activity.
Expand the sections below to learn more about M&M Rounds at Island Health or visit one of our M&M rounds subpages using the links at the bottom of the page.
- Already hosting M&M Rounds?
-
This section is for clinical groups already hosting M&M Rounds who are interested in transitioning to the new standardized approach.
The new model adapted by the HAMQC M&M Working Group asks that groups hosting rounds:
- Apply the principles of the Ottawa M&M model:
- Standardized approach
- Learning opportunity
- Protected discussion
- Medical education
- Quality improvement
- Risk management
- Identify the most appropriate Clinical Governance structure for reporting. This may a local-level clinical governance and/or C.A.R.E. committee
- Share the Bottom Line/Action item” slide with the designated Clinical Governance Structure (local-level and/or C.A.R.E. network
- Conduct case-based reviews
- Host rounds regularly
- Multidisciplinary involvement is recommended
- Focus lessons learned on cognitive bias and/or system factors
A key component of this model is encouraging inter-professional and multidisciplinary participation at M&M rounds. If it is not already happening, we recommend you open up invitations for M&M rounds to nursing and allied health professionals and other site representation, specialties or hospital administration where beneficial.
Island Health is also introducing “What Works” rounds as an opportunity to celebrate and share good practices and discuss the contributing factors. “What Works” rounds fit within the OM3 model and are a tool for summarizing clinical cases into tangible lessons or tips that can be shared broadly.
To learn more visit the M&M Rounds for Presenters page for detailed instructions on how to present rounds for your clinical group or read the Island Health M&M Toolkit for Presenters.
To learn more about “What Works” rounds visit the M&M Rounds “What Works” page.
- Apply the principles of the Ottawa M&M model:
- What do I need to know about Facilitating M&M Rounds?
-
Facilitators play a key role for successful M&M rounds by organizing and moderating rounds for your clinical group.
This includes:
- Ensuring regular rounds are scheduled – M&M Presentations can also form a component of other scheduled meetings. Section 51 of the BC Evidence Act can be applied to the M&M portions as long as the requirements for M&M rounds are met.
- Inviting inter-professional and multidisciplinary participation
- Engaging and preparing presenters
- Moderating and setting a blame-free environment
- Facilitating discussions
- Post-round reporting and follow up
The role of a facilitator is just as important post-rounds. Facilitators help to effect change by sharing bottom-line summaries with the participants at the rounds and with your designated Clinical Governance structure as identified for section 51 protection (this may be a local-level clinical governance and/or C.A.R.E. network structure).
Sharing issues and recommendations helps the organization to prioritize and action change, a concern identified locally may be echoed across the organization. Every voice counts.
To learn more visit the M&M Rounds for Facilitators for detailed instructions on how to facilitate rounds for your clinical group or read the Island Health M&M Overview Guide.
If you would like to schedule an onboarding session or learn more, please contact MedStaffQI@islandhealth.ca
- What should I know about Presenting M&M Rounds?
-
Presenters can drive improved quality of care and patient safety by sharing their experiences with adverse outcomes or near misses that contain lessons to be learned about cognitive bias and/or system factors using the structured M&M format.
Common cases that have the potential to occur frequently in your group’s practice setting present an opportunity for change.
The role of a presenter includes:
- Choosing an appropriate case that you were involved in. Your chosen case should include an adverse outcome or near miss and/or present an opportunity for learning or improvement.
- Performing a case analysis to determine any cognitive biases or system issues that may have contributed to the outcome.
- Creating ‘bottom lines’ that summarize cognitive and systems issues that could lead to action by your group.
- Presenting your case at M&M rounds.
- Working with the Facilitators to finalize bottom line summaries following discussion at rounds, including identifying potential system changes.
Presenters are also encouraged to share what went well using the “What Works” presentation format, celebrating good practices and highlighting contributing factors that can be shared broadly to improve quality of care.
To learn more visit the M&M Rounds for Presenters for detailed instructions on how to present rounds for your clinical group or read the Island Health M&M Overview Guide.
To learn more about “What Works” rounds visit the M&M Rounds “What Works” page.
- What should Attendees know about M&M Rounds?
-
Participation in M&M Rounds is a valuable way to improve patient quality of care and safety and is included in the Medical Staff Rules as an obligation of privileges.
As an attendee you are:
- Involved in identifying any system issues and cognitive biases
- Supporting a psychologically safe environment
- Learning under protection of Section 51 of the BC Evidence Act
- Obtaining CME Credits
All medical staff and multidisciplinary colleagues are eligible for membership of an M&M group. Ideally, all decisions will be taken by consensus. Meetings will begin and end on time. Psychological safety principles will apply. As a medical staff member of an M&M group you will be asked to take your turn at presenting a case of your choice that you have been involved in.
If you would like to learn more about attending M&M rounds please click here.
- What is different about this model compared to other M&M rounds?
-
M&M rounds focus on reviews for learning and serve a different purpose than critical incident reviews, case rounds, and medical grand rounds. M&M rounds can be utilized to support review of events reported in the Patient Safety and Learning System (PSLS), however, not all cases chosen for review at M&M rounds are patient safety events. Significant patient safety events (severe harm, death, and/or “Never Events”) may undergo reviews according to the Patient Safety Incident Management process, which also supports systems learning.
While M&M rounds support an approach to review events brought by patients and families either directly to a provider or through the Patient Care Quality Office (PCQO), they are separate from the PCQO process. To learn more click here and navigate to page 5 of the overview guide.
- At each M&M round, attendees will discuss the bottom-line slide developed by the presenter to identify cognitive biases or system issues that are suitable for action by your group to determine “what would you like to see happen?”.
- Action items discussed, whether they could be actioned locally or require a higher level of system change are shared with a designated Clinical Governance structure for learnings or as a driver for change. Sharing issues and recommendations helps the organization to prioritize and action change, a concern identified locally may be echoed across the organization.
A critical component for this model is encouraging inter-professional and multidisciplinary participation. A team-based approach to case discussion can offer important insights into not only the identification of cognitive and system issues related to a case, but also the development of potential solutions. Every voice counts!
To learn more about why Island Health has chosen the Ottawa Morbidity and Mortality Model (OM3) please expand the section “What is the Ottawa M&M Model?”
- What is the Ottawa M&M Model?
-
The Ottawa Morbidity and Mortality Model (OM3) model was chosen at Island Health because it focuses on reviews for learning and serves a different purpose than critical incident reviews, case rounds, and medical grand rounds.
Key components of the OM3:
- Standardized approach
- Learning opportunity
- Protected discussion
- Medical education
- Quality improvement
- Risk management
More information about why Island Health has chosen to adapt and adopt the OM3 framework can be found here. The guide includes detailed information on how to present and facilitate M&M rounds. You can also visit the sub-pages for Presenters and Facilitators on the Medical staff website.
Cases selected for M&M Rounds require all three of the following:
- Adverse outcome such as death, disability, harm, injury or a near miss (potential harm avoided).
- Preventable.
- Lessons to be learned about cognitive bias and/or system issues.
This format is different from a critical incident analysis and when using the model, cases are presented by those who were involved directly, not reviewed by a third party.
M&M rounds following the model are typically 30 minutes and include:
- 10 minutes for review of the case and state of evidence on current management
- 10 minutes for case analysis in terms of cognitive and system issues
- 10 minutes for discussion, review of bottom lines and consensus on potential action items.
The OM3 model also encourages “What Works” rounds as an opportunity to celebrate and share good practices and discuss the contributing factors. “What Works” rounds are a tool for summarizing clinical cases into tangible lessons or tips that can be shared broadly. To learn more about “What Works” rounds visit the M&M Rounds “What Works” page.
- What happens after rounds?
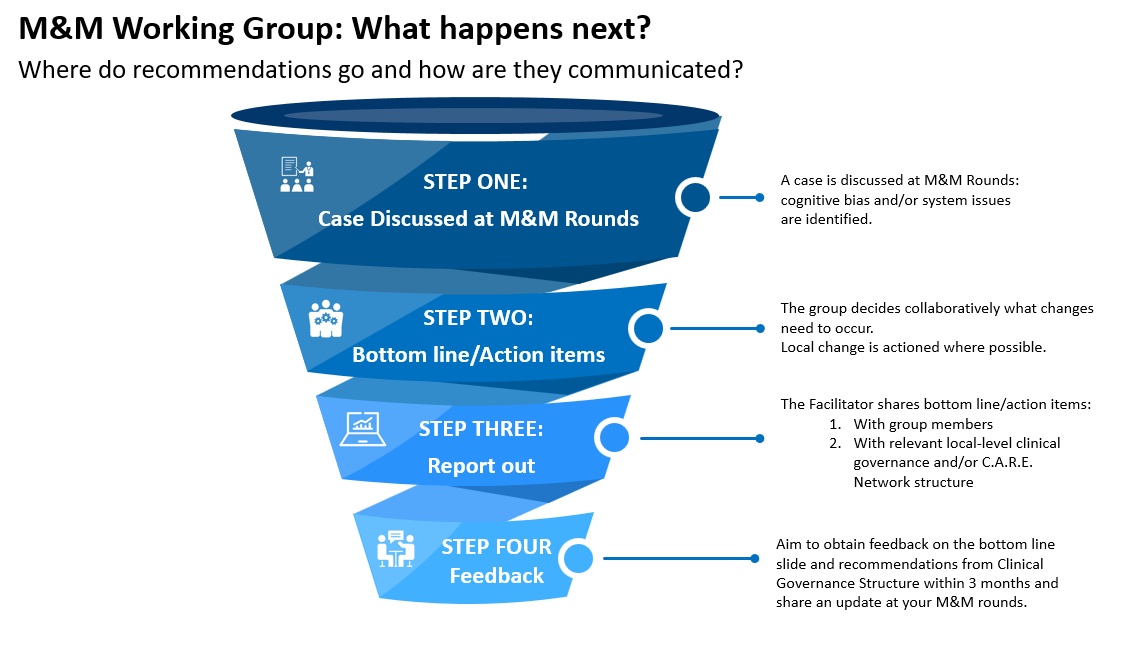
Efforts to improve the impact of your M&M rounds do not end once the presentations are over.
Each M&M round should end with discussion around potential ideas for change and concrete action items. This information, captured on the bottom line slide, is then shared with your most appropriate clinical governance structure. This may be your relevant local-level clinical governance and/or C.A.R.E. Network structure.
Aim to obtain feedback on the bottom line slide and recommendations from the designated Clinical Governance Structure within 3 months. The facilitator will then share this information as an update at rounds – the cycle of change continues!

M&M Rounds: What happens next? - M&M Rounds Data Reporting
-
Each M&M round should end with discussion around potential ideas for change and concrete action items. This information, captured on the bottom line slide, is then shared with your most appropriate clinical governance structure. This may be your relevant local-level clinical governance and/or C.A.R.E. Network structure.
Aim to obtain feedback on the bottom line slide and recommendations from the designated Clinical Governance Structure within 3 months.
- M&M Rounds and Medical Staff Rules
-
As outlined in the Medical Staff Rules, 2.2.4.4 “Department members will participate in a program of structured quality assurance, including Morbidity and Mortality rounds and case reviews arising from quality committee activities or complaints, regarding the care provided to patients by its Members.”
- Introducing “What Works Rounds” for celebrating and sharing success
-
“What Works” (WW) or “What Went Well” rounds provide an opportunity to celebrate and share good practices using the OM3 framework. The aim is to summarize clinical cases into tangible lessons or tips that can be shared widely to improve quality of care, emphasizing the contributing factors whether they are individual, teamwork, or environmental/system factors.
WW rounds follow a similar format to presenting M&M rounds, though the case selection and areas of focus differ from the traditional format.
To learn more about WW rounds visit the “M&M Rounds “What Works” page. More details on how to choose a case and perform a case analysis for WW rounds can be found in the Island Health Guide to “What Works” Rounds.
An overview of the new standardized M&M Rounds model

Click here to view a PDF of the Cycle of Change.